
We show that 19th century atmospheric levels, before significant anthropogenic influence, were stable at We find a significant peak in CF4 and C2F6 emissions aroundmost likely due to the high demand for aluminium during World War II, for example for construction of aircraft, but these emissions were nevertheless much lower than in recent years. We present an inversion for global emissions from to We also formulate the inversion to directly infer emission factors for PFC emissions due to aluminium production prior to the s. We take account of the age characteristics of the firn and ice core air samples and demonstrate excellent consistency between the ice core, firn and atmospheric measurements. Perfluorocarbons PFCs are very potent and long-lived greenhouse gases in the atmosphere, released predominantly during aluminium production and semiconductor manufacture. Stay up to date with the latest developments. Ask a New Question.Our leaders are working closely with federal and state officials to ensure your ongoing safety at the university. Using data the boiling point constants given in the test book, calculate asked by m on February 15, CHEM 2: Boiling Point Which Aqueous solution has the highest normal boiling point? The answer I found was that: asked by Sara on December 6, You can view more similar questions or ask a new question. Questions chem class which of the following has the highest boiling point? Chemistry Arrange the following aqueous solutions in order of increasing boiling point, explain your answer: 0. You can view more similar questions or ask a new question. Explain the difference in their boiling points. Which of the following compounds has the highest boiling point? Diethyl ether has a boiling point of Both of these compounds have the same numbers and types of atoms. I think that the answer is D, the all have the same boiling point. Which Aqueous solution has the highest normal boiling point? They all have the same boiling point.
Using data the boiling point constants given in the test book, calculate.

Which of the following molecules has the highest boiling point? Arrange the following aqueous solutions in order of increasing boiling point, explain your answer: 0. Arrange the compounds in order of increasing boiling point. The Molecule with the highest molecular weight has stronger intermolecular dispersion forces. Now it all comes to the Dispersion Forces. The examples above have no hydrogen bonds. Hydrogen bonds only exist when there are hydrogen atoms bonded with one of three: Fluorine, Oxygen or Nitrogen. Since the molecules are proven non-polar, there are no permanent dipole-dipole interactions between them. A molecule may contain polar bonds but they cancel each other and the overall molecule becomes non-polar. It all depends on the type and strength of the intermolecular forces between the molecules. So the one with the highest molar mass will be the highest boiling point. Community Guidelines.Intermolecular forces play a role in boiling points, for sure, but in this case it's a matter of molecular weight. Is BrF5 polar or non-polar? It is nonpolar. Is CH3Cl polar or non polar? Yes it is polar.
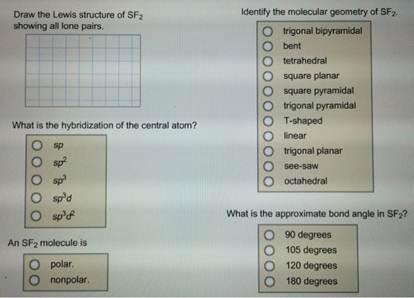
#Sf2 polar or nonpolar code#
What virtual families crack code and name? Is soap polar or non-polar? Soap is polar and non-polar. Wiki User C2F2 is nonpolar because the polar bonds cancel out. The material on this site can not be reproduced, distributed, transmitted, cached or otherwise used, except with prior written permission of Multiply. What are the characteristics of the various materials that are used with regard to the bending operations and why do some materials require to the addition of heat to aid the bending process? All Rights Reserved. Is best defined as the total weight of persons gear equipment stores fuel and motor assembly found on a vessel? Have you ever crashed a wedding or had your wedding crashed, if so what happened? Mrs Smith has nine children half of them are girls.

The molecule would have a quadrupole moment, but I don't know the value.
